
US regulators have already granted 'emergency use' authorisation for three Covid-19 vaccines
- This article is more than five years old.
- Published on May 11, 2021 at 16:05
- Updated on May 14, 2021 at 15:55
- 3 min read
- By Tendai DUBE, AFP South Africa
A Facebook post shared in South Africa and the United States has attracted more than 2,900 views and hundreds of shares since its publication on April 29, 2021.
It includes a 55-second video clip showing former FDA commissioner Stephen Hahn testifying before the US Congress about Covid-19 vaccines, and assuring Americans that the agency would only approve those that have undergone rigorous testing.
The claim appears as a line of text superimposed on the video and reads: “LISTEN CAREFULLY FOLKS. FDA ‘REFUSING’ TO APPROVE VAX’S. Pray for those who have already taken it! I’M SHOCKED! (sic)”.
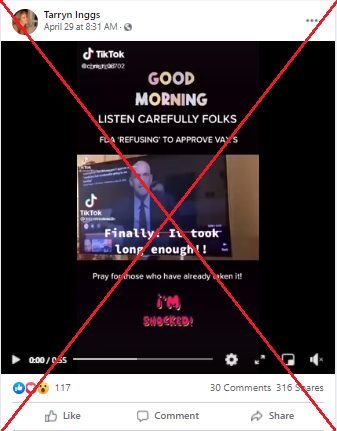
Hahn had testified the agency would never approve a vaccine that its own officials felt uncomfortable giving to their families.
“On behalf of 17,000-plus employees of the FDA, I want to make the following commitments to the American public and this committee: the FDA will not authorise or approve any Covid-19 vaccine before it has met the agency’s rigorous expectations for safety and effectiveness,” he said.
The claim, however, distorts his words.
At no point in the video did Hahn give the impression that the FDA would deny approval for any vaccines. Furthermore, the testimony was filmed on September 23, 2020, before any Covid-19 vaccines had been approved in the US.
Currently, the FDA has authorised three vaccines for “emergency use” in the United States, made by Pfizer-BioNTech, Moderna, and Janssen.
An Emergency Use Authorization (EUA), according to the FDA, is used to “facilitate the availability and use of medical countermeasures, including vaccines, during public health emergencies, such as the current COVID-19 pandemic.”
The authority also further extended the use of the Pfizer-BioNTech vaccine for 12-to-15-year-olds on May 11, 2021.
FDA on vaccines
The excerpt was taken from a three-hour-long testimony on pandemic response efforts on September 23, 2020, by White House Coronavirus Task Force members.
According to the summary on US television network C-Span, “all four witnesses expressed strong confidence in the scientific review process for which vaccine candidates would be considered for emergency authorisation use approval.”
The testimony was recorded before any emergency use authorisations had been granted and amid heated debate over whether former US President Donald Trump was pushing more strongly for vaccines ahead of the November 2020 election.
At the time, scientists were actively trying to allay fears that the approval process was giving in to political pressure.
In the full video, just before the segment shared on social media, Hahn had said: “When a vaccine sponsor reaches the conclusion that the data from its Phase 3 clinical trials are adequate to submit to FDA, they will decide whether to apply for approval or Emergency Use Authorisation.”
He said this would be based on the trial meeting “pre-specified success criteria that were established by that sponsor,” but most importantly, they should also be aligned with FDA recommendations for those criteria.
“FDA will receive that application, or submission, and our career scientists will review it, safety and efficacy data, as well as manufacturing quality and consistency data,” Hahn said.
In updated testimony from federal officials on May 11, 2021, Peter Marks, director at the Center for Biologics Evaluation and Research, said that the FDA expected the manufacturers whose Covid-19 vaccines have been authorised for emergency use “to continue their clinical trials” in order to obtain full approval.
“The three authorisations make available Covid-19 vaccines in the United States that have shown clear and compelling effectiveness in large, well-designed Phase 3 trials and that meet rigorous standards for safety and effectiveness to support emergency use authorisation,” said Marks.
Vaccine hesitancy
The claim is part of a flood of misinformation about vaccines that continues to spread online as nations seek to immunise people against Covid-19.
More than 3,346,813 people have died since the start of the pandemic.
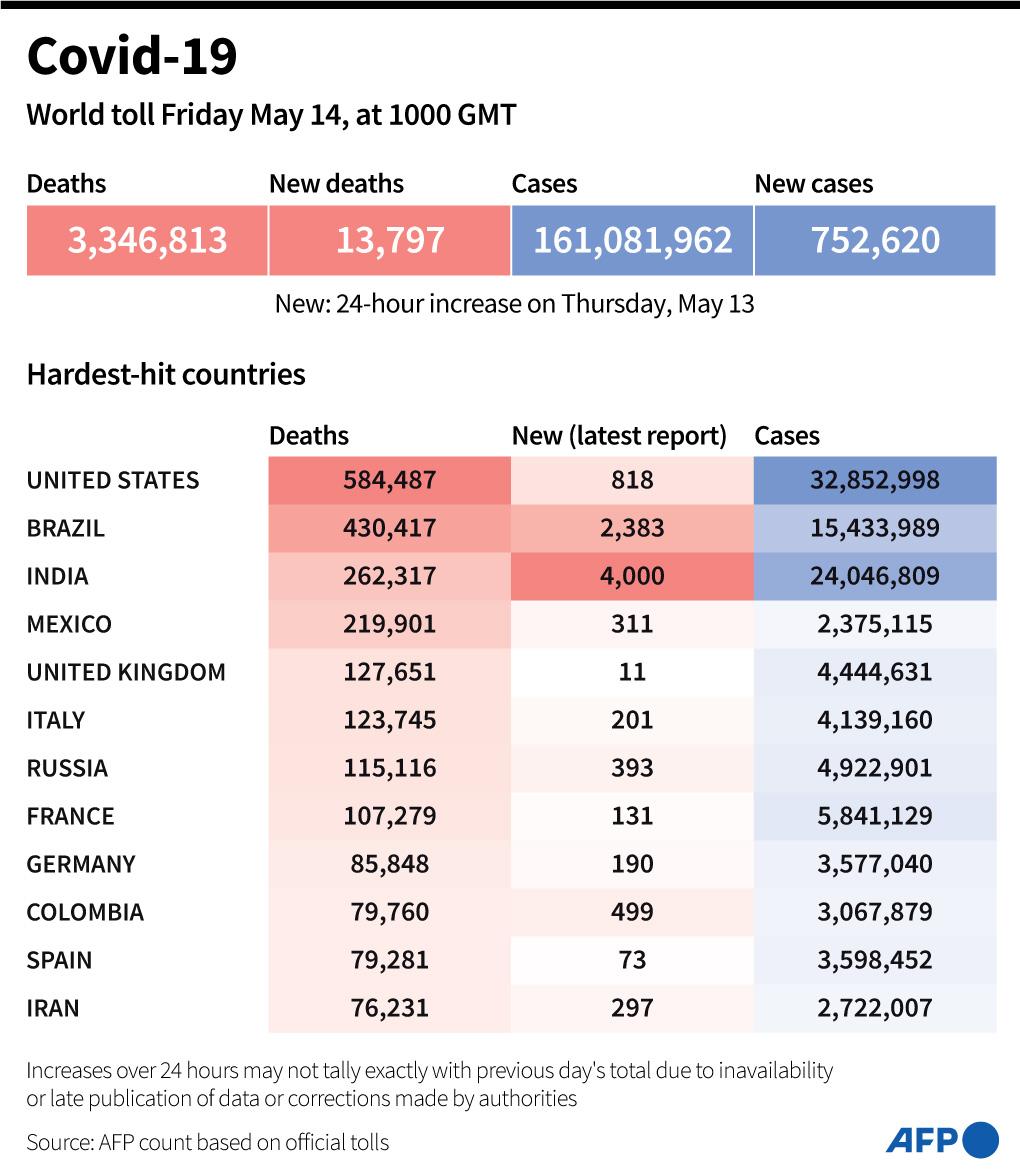
Worldwide, at least 1.377 billion doses of Covid-19 vaccines have been administered, in at least 210 countries or territories, according to an AFP tally based on official sources at 08:58 GMT on May 13, 2021.

Copyright © AFP 2017-2026. Any commercial use of this content requires a subscription. Click here to find out more.
Is there content that you would like AFP to fact-check? Get in touch.
Contact us
