US health agencies are not scrapping common coronavirus test
- This article is more than four years old.
- Published on July 29, 2021 at 20:49
- 3 min read
- By Claire SAVAGE, AFP USA
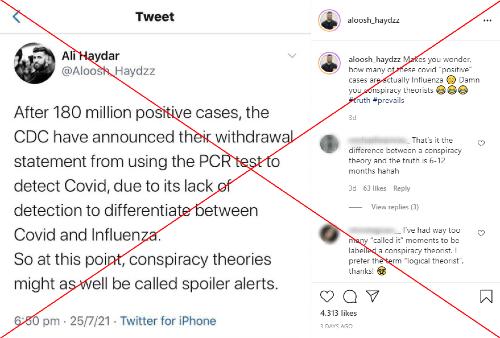
"After 180 million positive cases, the CDC have announced their withdrawal statement from using the PCR test to detect Covid, due to its lack of detection to differentiate between Covid and Influenza," says a July 25, 2021 Instagram post, referring to polymerase chain reaction (PCR) tests.
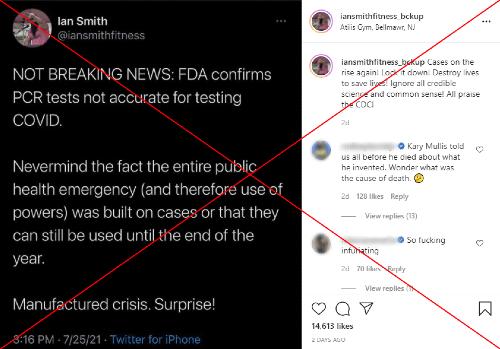
Another Instagram post, liked about 14,600 times, shared a screenshot of a tweet saying: "NOT BREAKING NEWS: FDA confirms PCR tests not accurate for testing COVID. Nevermind the fact the entire public health emergency (and therefore use of powers) was built on cases or that they can still be used until the end of the year. Manufactured crisis. Surprise!"
Similar claims appeared on Facebook here, here and here and in an online article here.
PCR tests are used by health care workers around the world to detect SARS-CoV-2, the virus that causes Covid-19, and have been a regular target of misinformation during the pandemic.
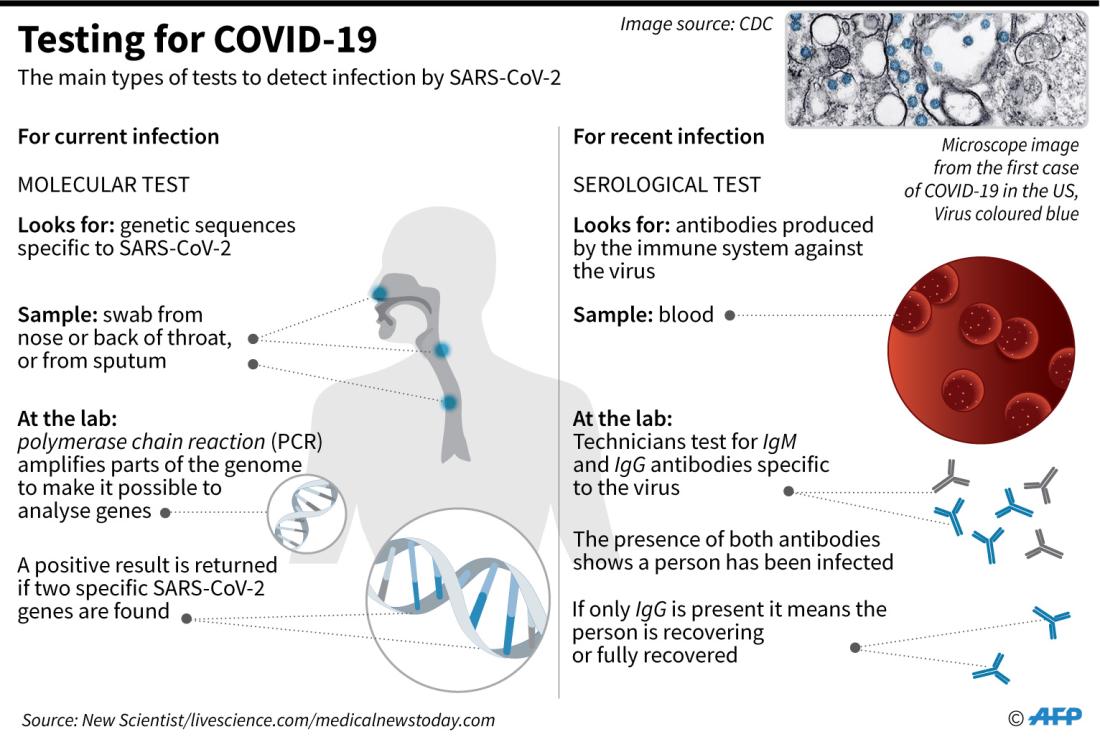
Many tests for Covid-19 are approved, and the Centers for Disease Control and Prevention (CDC) is seeking to discontinue just one of them.
The organization said it will withdraw a request for Emergency Use Authorization (EUA) of an RT-PCR test it developed at the beginning of the pandemic, CDC spokeswoman Jade Fulce explained.
But the move was not due to performance issues. Instead, demand for the CDC's test declined with the emergence of higher-tech procedures, she said.
Several posts draw on language from the CDC's announcement of that withdrawal to falsely claim PCR tests cannot tell the difference between Covid-19 and the flu.
The announcement said the CDC "encourages laboratories to consider adoption of a multiplexed method that can facilitate detection and differentiation of SARS-CoV-2 and influenza viruses."
But Fulce said the agency was referring to tests that can identify both SARS-CoV-2 and flu viruses at the same time, and that it does not mean past tests conflated the two.
The posts echo Covid-19 skeptics who have pushed the false narrative that the CDC added flu cases and deaths to those of the coronavirus in order to make the pandemic appear more deadly.
Jim McKinney, a spokesman for the US Food and Drug Administration (FDA), told AFP on July 27 that PCR tests are considered the best way to test for Sars-CoV-2 .
"The FDA has not issued any statement questioning the reliability of PCR test results in general," he said.
"To date, the FDA has authorized more than 300 tests and sample collection kits to diagnose Covid-19, many of which are PCR tests. PCR tests are generally considered to be the 'gold standard' for Covid-19 diagnosis," McKinney added.
Ana Santos Rutschman, assistant professor at St Louis University's Center for Health Law Studies, said that EUAs are meant to be temporary.
"A revocation in itself is just a technical means to giving the product a different status... the CDC is endorsing a different type of test, but not saying anything about PCR tests" in general, Rutschman said.
Jennifer Piatt, a researcher at the Arizona State University Center for Public Health Law and Policy, agreed.
"Nothing in the CDC's statement indicates that its test conflates Covid-19 with influenza, or that PCR tests in general do so. Arguments to the contrary misstate the CDC's language," she said.
Instead, "the statement simply indicates CDC's support for tests that can detect both influenza and COVID-19 for purposes of saving time and resources," said Piatt, who is also a senior attorney with the Network for Public Health Law.
AFP Fact Check has debunked other inaccurate claims about PCR tests here.
Copyright © AFP 2017-2026. Any commercial use of this content requires a subscription. Click here to find out more.
Is there content that you would like AFP to fact-check? Get in touch.
Contact us