Philippine authorities warn anti-viral injection has not yet been approved for treating COVID-19
- This article is more than six years old.
- Published on April 15, 2020 at 09:30
- 1 min read
- By AFP Philippines
This Facebook post shared the claim on April 8, 2020.
The post shows a stock photo of a person holding a medical syringe overlaid with text that reads: “FAI / Fabunan AntiViral Injection is the CURE for COVID-19”.
Below is a screenshot of the misleading post:
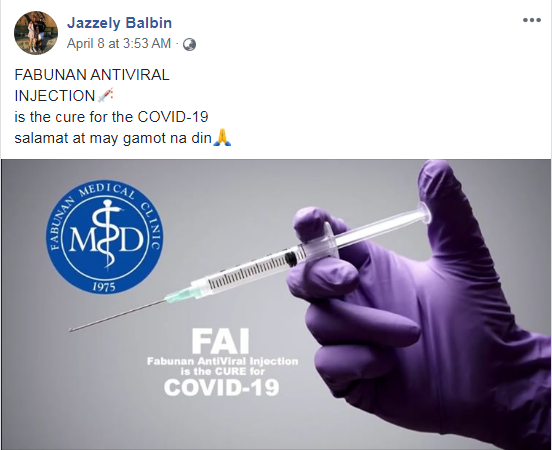
The post’s caption, written in a mix of English and Tagalog, reads: “FABUNAN ANTIVIRAL INJECTION / is the cure for the COVID-19 / thanks and there’s a cure”.
The same photo was also shared alongside a similar claim on Facebook here, here, here and here.
The claim is misleading.
The Philippine FDA, which regulates medical and pharmaceutical standards in the country, warned the public against using the purported injection, saying the injection's effectiveness in treating COVID-19 was “unproven”.
“Currently there are no registered drugs or vaccines that are licensed specifically for use in COVID-19 treatment or prevention,” the agency said in this statement on April 8, 2020.
“Manufacturers and vendors of unlicensed products such as PRODEX B and the Fabunan Antiviral Injections have been informed regarding the process and requirements to register their products with the FDA Center for Drug Regulation and Research (CDRR),” it said.
“No applications for product registration have been filed to date. Until such products are proven safe and effective for use in the treatment of COVID-19 they cannot be dispensed to the public nor can therapeutic claims be made.”
“Prodex B” is an unregistered anti-COVID-19 drug combination that the FDA had earlier warned against, AFP reported here.
As of April 15, 2020, the FDA has not updated its position on the Fabunan Antiviral Injections.
The WHO also maintains here there is currently no vaccine or antiviral medicine that has been proven to prevent or treat COVID-19 as of April 2020.
The disease, first detected in the central Chinese city of Wuhan in late 2019, has killed more than 117,000 people and infected at least 1.8 million globally as of April 14, 2020, WHO figures show.
Copyright © AFP 2017-2026. Any commercial use of this content requires a subscription. Click here to find out more.
Is there content that you would like AFP to fact-check? Get in touch.
Contact us