The Covid-19 test kits are not for use with water, manufacturer says
- This article is more than four years old.
- Published on July 6, 2021 at 09:51
- Updated on July 7, 2021 at 04:14
- 3 min read
- By AFP Malaysia
The video was posted on June 21, 2021, on Facebook here, where it has been viewed more than 7,400 times.
The two-minute, 43-second video shows someone tearing open a Covid-19 rapid test device from Abbott, testing it with water straight from the tap, and receiving two lines, which indicate a positive result.
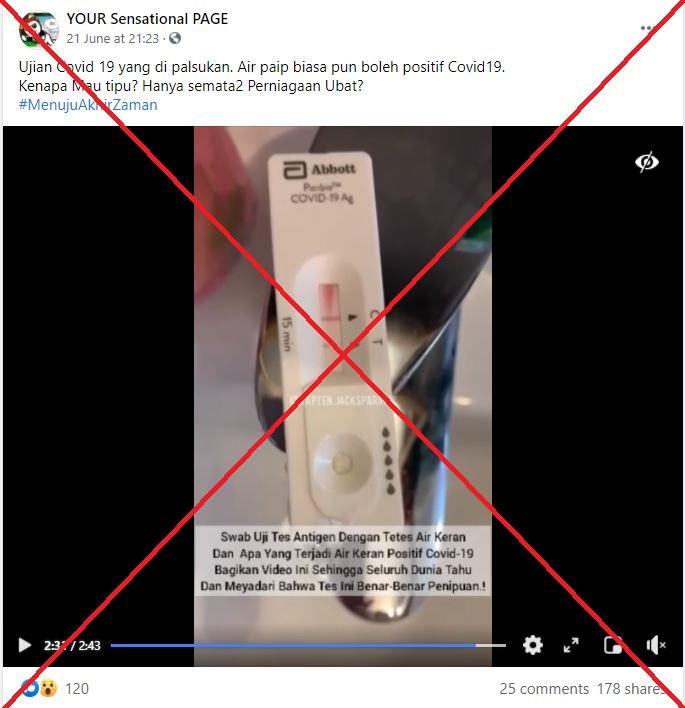
The post’s Malaysian-language caption translates to English as: “Covid-19 tests are faked. Tap water can test positive for Covid-19.
“Why do they want to deceive? Only for pharmaceutical business?”
The text superimposed on the video reads: “Antigen swab test with tap water and what happens tap water is positive for Covid-19
“Share this video so the whole world know and realise that this test is really a scam!”
Abbott Laboratories is a US-based medical devices and health care company. It is one of a number of companies that manufacture rapid test kits to detect Covid-19.
The video was also shared with a similar claim here.
It was also posted with a similar claim in various languages, such as in English on Facebook here, on Twitter here and on BitChute here.
It also appeared with a similar claim in Indonesian on Twitter here and in Hindi on YouTube here.
The claim is false as the Covid-19 test kit was not used correctly.
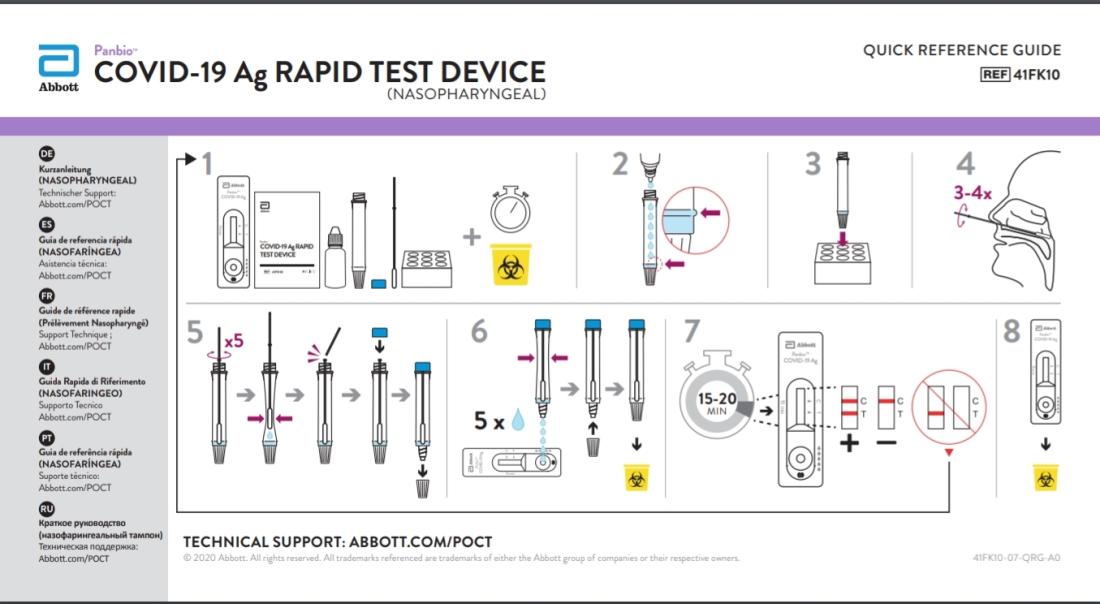
A closer look at the video found that the rapid test device used by the person is a Panbio COVID-19 Antigen Rapid Test Device produced by Abbott. The test device is for professional use only and for use with nasal or nasopharyngeal swab samples.
Contacted by AFP on June 28, 2021, an Abbott spokesperson said: “Panbio COVID-19 Ag Rapid Test Device is for use with samples collected with a nasal or nasopharyngeal swab inserted into a person’s nostrils. Panbio Ag is not for use with water or any other foods or liquids.
“When used as intended, it is a highly accurate test that is helping to detect COVID-19 across the world and can significantly improve efforts to control transmission.
“Spreading misinformation with deliberate misuse of a medical product during a pandemic is misleading and irresponsible.”
Here is a reference guide from Abbott, published on the World Health Organization’s website, on how to use the rapid test device.
Here is a video instruction from the Provincial Health Services Authority ( (PHSA), the health authority in British Columbia province, Canada.
Other examples of general users using the rapid test device from Abbott can be found on Singaporean media The Straits Times and Channel News Asia.
In Malaysia, antigen rapid test kits (as well as antibody rapid test kits) can only be used by qualified and trained health care professionals as they are regulated under the Medical Devices Act 2012.
The US Centers for Disease Control and Prevention (CDC) has this guideline for antigen testing, which says: “Quality assurance procedures should be followed to prevent cross-contamination and inaccurate test results. For example, users should follow the manufacturer’s instructions, as well as state and local guidance, for when and how often to perform testing on control specimens.”
AFP previously fact checked other misleading claims on rapid tests for Covid-19 here, here, here and here.
Other items that purportedly tested positive for Covid-19 in these false claims include the flu shot and Coca-Cola.
Copyright © AFP 2017-2026. Any commercial use of this content requires a subscription. Click here to find out more.
Is there content that you would like AFP to fact-check? Get in touch.
Contact us