Misleading claim circulates in Burmese about WHO ‘designating’ Russia’s Covid-19 vaccine as official inoculation
- This article is more than five years old.
- Published on November 11, 2020 at 06:00
- 2 min read
- By AFP Myanmar
The claim was published here on a Burmese-language website on November 3, 2020.
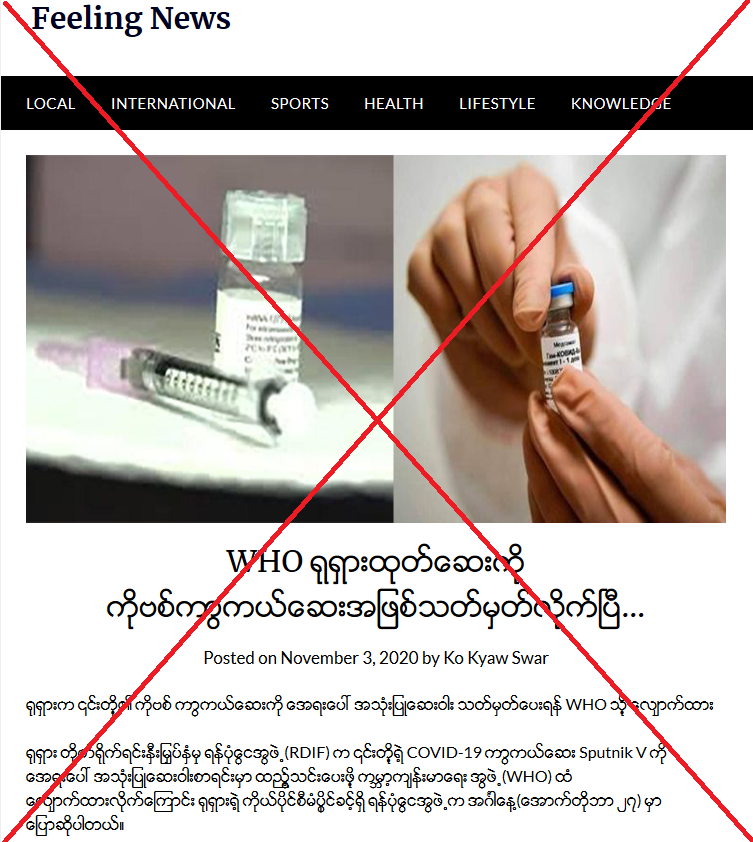
The title of the Burmese-language post translates in English as: “WHO has designated Russian made vaccine as COVID-19 vaccine”.
After the misleading headline, the article says: “Russia applies to the WHO for emergency use of its vaccine.
“Russia's Direct Investment Fund (RDIF) has applied to the World Health Organization (WHO) for inclusion of its COVID-19 vaccine Sputnik V in for Emergency Use Listing, Russia’s sovereign wealth fund said on (October 27) Tuesday.”
The article was widely shared on Facebook, for example here, here, here, and here.
However, the claim in the headline is misleading.
The WHO has not designated any Covid-19 vaccine and says on its website that “there are no medicines that have been shown to prevent or cure the disease”.
It also says clearly on its website that there is no current vaccine: “To date, there is no vaccine and no specific antiviral medicine to prevent or treat COVID-2019.”
On October 27, 2020, the Russian Direct Investment Fund, which oversees the country’s vaccine production, published this statement about its application to the WHO for an Emergency Use Listing of the country’s Sputnik V coronavirus vaccine.
The press statement states in part: “The Russian Direct Investment Fund has submitted applications to the World Health Organization for accelerated registration (Emergency Use Listing, EUL) and prequalification of the world’s first registered vaccine against the coronavirus Sputnik V, which is based on a well-studied platform of human adenoviral vectors.”
This registration had not been granted as of November 11.
Multiple Covid-19 vaccines are being trialed and on November 11 US pharmaceutical giant Pfizer said its drug had shown 90 percent effectiveness, sending markets soaring and raising hopes of an end to the worst pandemic in a century. Here is an AFP report on the development.
WHO Director-General Tedros Adhanom Ghebreyesus hailed the news as "encouraging" shortly after warning that the world "might be tired of Covid-19. But it is not tired of us."
Copyright © AFP 2017-2026. Any commercial use of this content requires a subscription. Click here to find out more.
Is there content that you would like AFP to fact-check? Get in touch.
Contact us