Indian social media posts, media reports falsely claim WHO approved herbal medicine as Covid-19 treatment
- This article is more than five years old.
- Published on February 26, 2021 at 10:10
- 2 min read
- By AFP India
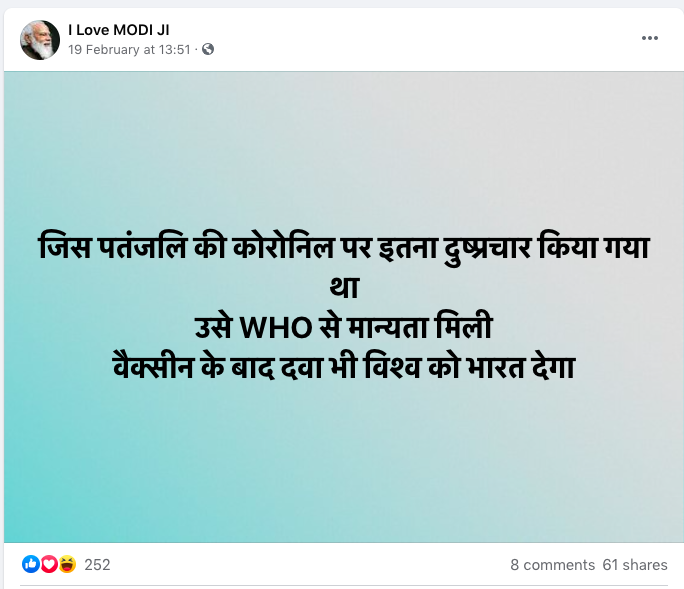
The claim was published in this Facebook post on February 19, 2021.
The post's Hindi-language text translates to English as: “Patanjali's coronil, which was targeted by negative propaganda, has been approved by the WHO. After the vaccine, India will lend the first Covid drug to the world.”
Patanjali is an Indian manufacturer of herbal products — including Coronil, which the company describes as a “Covid-19 Immunity Booster Tablet.”
The claim that the WHO approved Coronil as a Covid-19 treatment was also made by News Nation TV. One of the Indian broadcaster's network anchors reported here that “a lot of questions were raised whether Coronil is an effective treatment of coronavirus, but WHO has given its approval.”
Several other media Indian outlets — including India TV; AsiaNews; Jio News; and News18 — also reported that the WHO had approved Coronil as a coronavirus treatment.
A similar claim was also shared on Facebook here, here and here. Sanju Verma, a national spokesperson for India’s ruling Bharatiya Janata Party, promoted the claim on Twitter here.
The claim, however, is false.
The WHO’s regional office for Southeast Asia said it had not approved the purported coronavirus treatment.
“WHO has not reviewed or certified the effectiveness of any traditional medicine for the treatment #COVID19,” the agency said on Twitter here on February 19, 2021.
.@WHO has not reviewed or certified the effectiveness of any traditional medicine for the treatment #COVID19.
— WHO South-East Asia (@WHOSEARO) February 19, 2021
On February 19, 2021, Patanjali’s managing director Acharya Balkrishna clarified in a post on Twitter that Coronil had received approval from the Indian government, not from the WHO.
The tweet reads: “We want to clarify to avoid confusion that our WHO GMP compliant COPP certificate to Coronil is issued by DCGI, Government of India. It is clear that WHO do not approve or disapprove any drugs. WHO works for building a better, healthier future for people all over the world".
DCGI is an acronym for the Drugs Controller General of India, who heads the government’s Central Drugs Standard Control Organization.
COPP, which is often written as CPP, is an acronym for Certificate of Pharmaceutical Product, which are uniform certificates used for international trade.
Balkrishna’s clarification came one day after Patanjali published a tweet claiming the WHO had approved Coronil. The tweet was deleted but an archived version can be seen here.
We want to clarify to avoid confusion that our WHO GMP compliant COPP certificate to Coronil is issued by DCGI, Government of India.
— Acharya Balkrishna (@Ach_Balkrishna) February 19, 2021
It is clear that WHO do not approve or disapprove any drugs.
WHO works for building a better, healthier future for people all over the world. pic.twitter.com/ZEDPdWy0tg
In a statement released on February 22, 2021, the Indian Medical Association, a voluntary organisation of Indian doctors, criticised Patanjali for promoting the false claim, calling it a “blatant lie of WHO certificate for a secret medicine.”
Coronil, however, did recently receive a CPP from the Indian government’s Central Drugs Standard Control Organisation, as Balkrishna noted in his tweet above and as the Press Trust of India reported here.
Copyright © AFP 2017-2026. Any commercial use of this content requires a subscription. Click here to find out more.
Is there content that you would like AFP to fact-check? Get in touch.
Contact us