
South Korea has not proposed legislation to make PCR tests mandatory
- This article is more than four years old.
- Published on September 8, 2021 at 11:40
- 2 min read
- By Richard KANG, AFP South Korea
The claim was shared here on South Korean blogging platform Naver Blog on August 21, 2021.
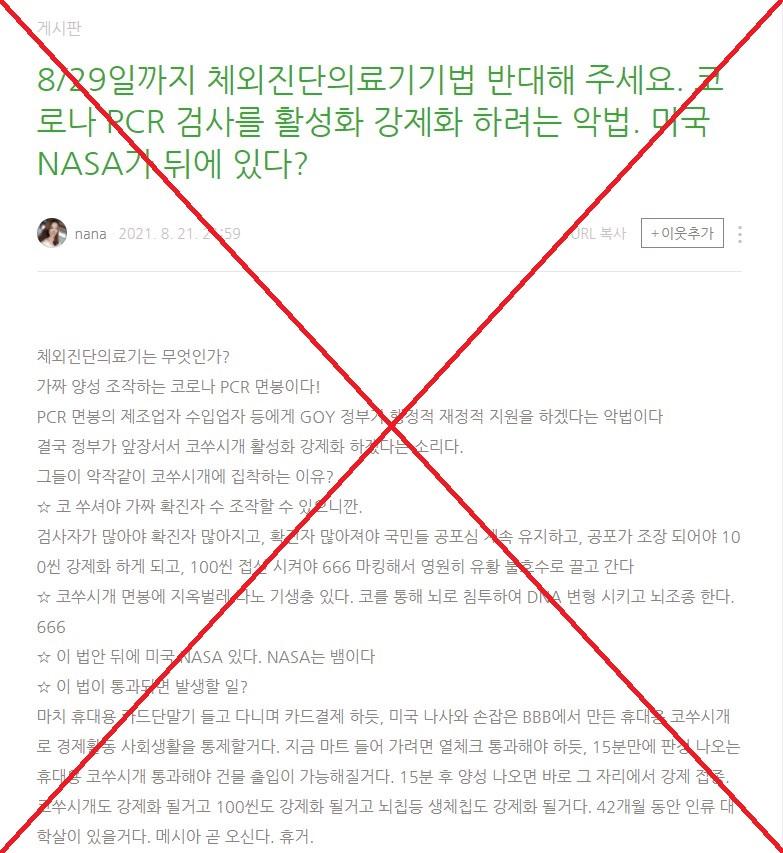
Part of the Korean-language claim translates to English as: "The proposed bill is designed to subsidise PCR swab makers and importers. It is an evil law. The government is taking the lead to make PCR tests mandatory and manipulate the number of the confirmed cases."
The post cites a bill proposed on August 19, 2021 by a group of Seoul-based lawmakers led by Choi Hye-young from the Democratic Party of Korea.
If passed, it would give the South Korean government greater powers to regulate manufacturers and importers of in vitro diagnostic medical devices.
The bill reads in part: "The bill aims to ensure that in vitro diagnostic medical devices manufacturing, importing, and performance tests are safely carried out and to protect the public health".
The post also shared a screenshot of the proposed bill's front page.
The superimposed text reads: "The law to make PCR tests mandatory".

A similar claim was shared on Facebook here, here and here.
However, the claim is misleading.
A spokesperson for Choi's office said there was no intention to make PCR tests mandatory with the proposed legislation.
"We would like to stress that there are no mentions of making PCR tests compulsory in the bill or elements that could lead this bill to be used for such practice. There is also no intention of doing so behind this bill," the official told AFP via email on September 7, 2021.
AFP found that as of September 8, the bill includes no reference to making PCR tests mandatory.
In fact, it focuses on regulating the manufacturers and importers of in vitro diagnostic medical devices.
The spokesperson for Choi's office continued: "We learnt that several leading economies around the world set up certain standards to ensure the quality of in vitro diagnostic medical devices, including having manufacturers to equip with facilities to prevent product contamination.
"However, many manufacturers of such devices in South Korea operate on a small scale, which often means they do not have capabilities to prepare these facilities. Given that, we've decided to support them with this new bill".
Copyright © AFP 2017-2026. Any commercial use of this content requires a subscription. Click here to find out more.
Is there content that you would like AFP to fact-check? Get in touch.
Contact us
